
1 / 5











| Product Name | Transparent IV Dressing (A0607MU) |
|---|---|
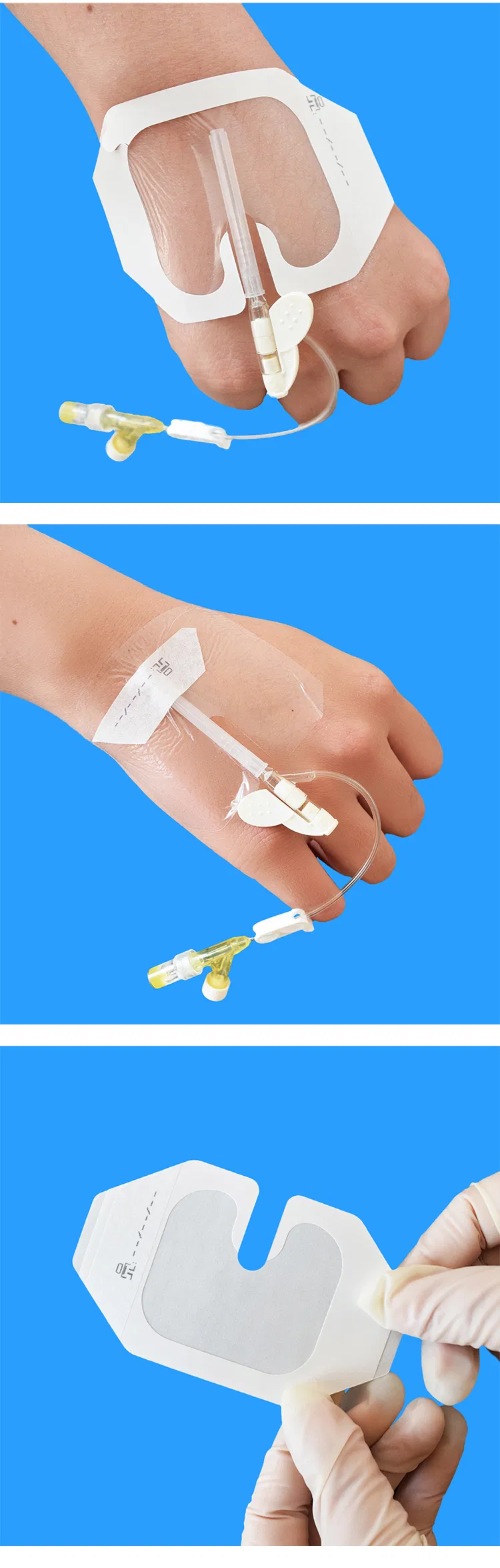
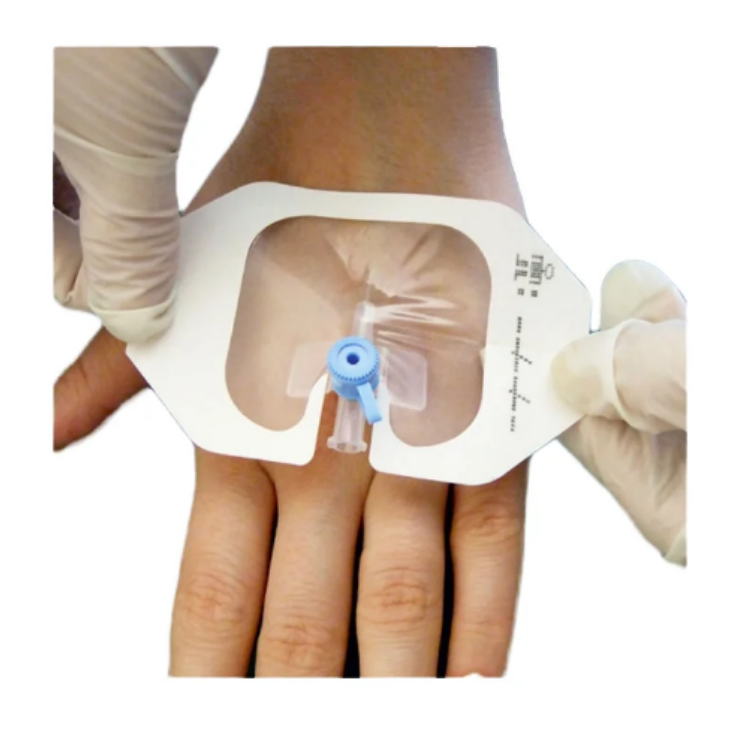
| Features | Hypoallergenic, Sterile, Air Permeable, X-Ray Permeable, No Residue |
| Application | Hospitals, Clinical Centers, and Rehabilitation Facilities |
| Specification | 6cm*7cm, PU film transparent with release paper |
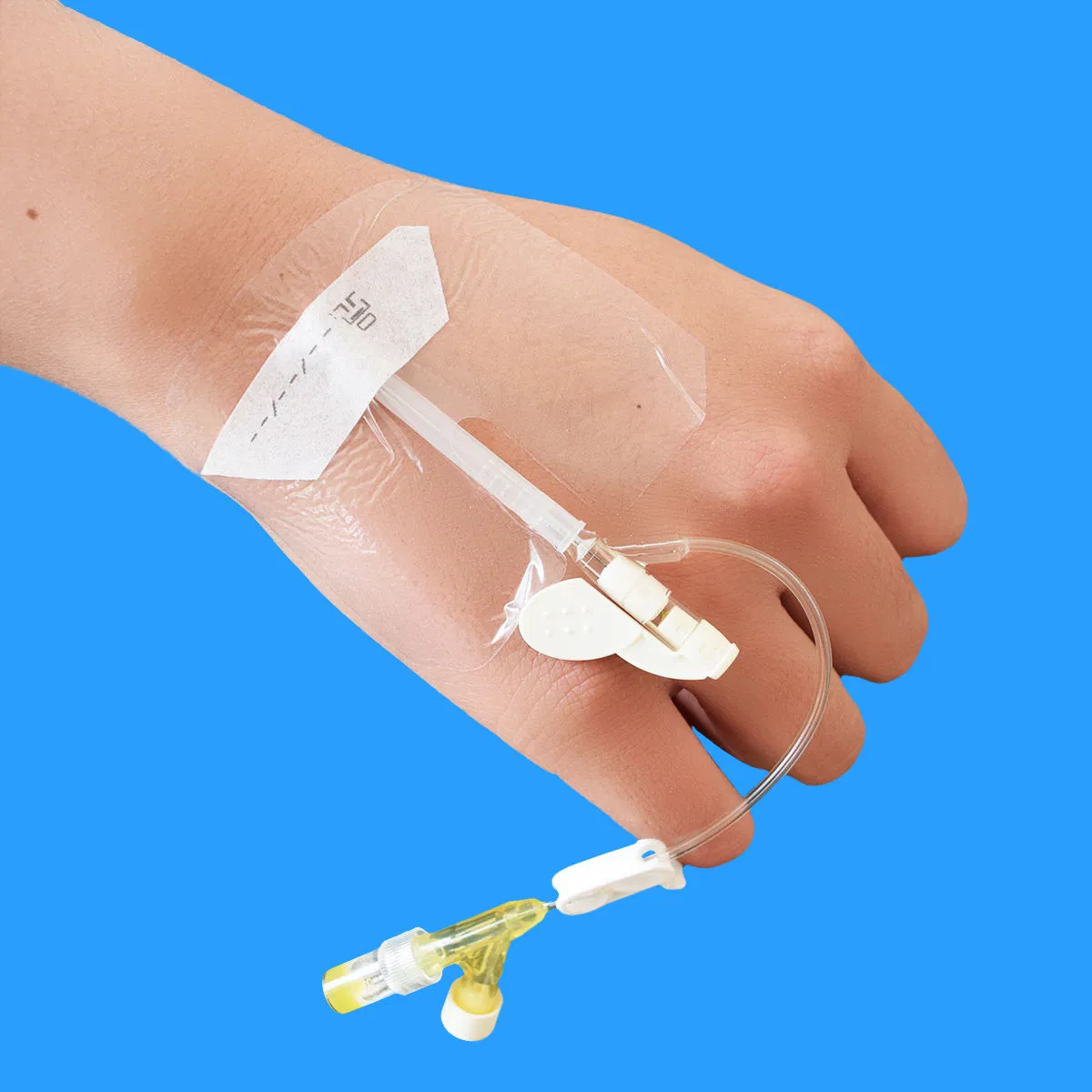
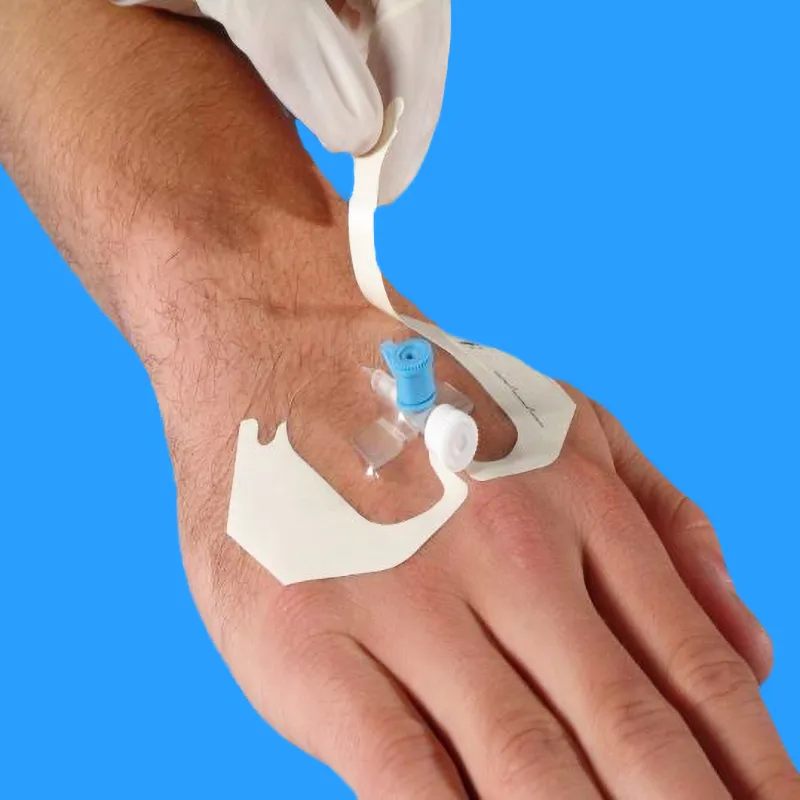
| Clinical Benefit | Excellent cannula stability allows patients to move freely while protecting the insertion site. |

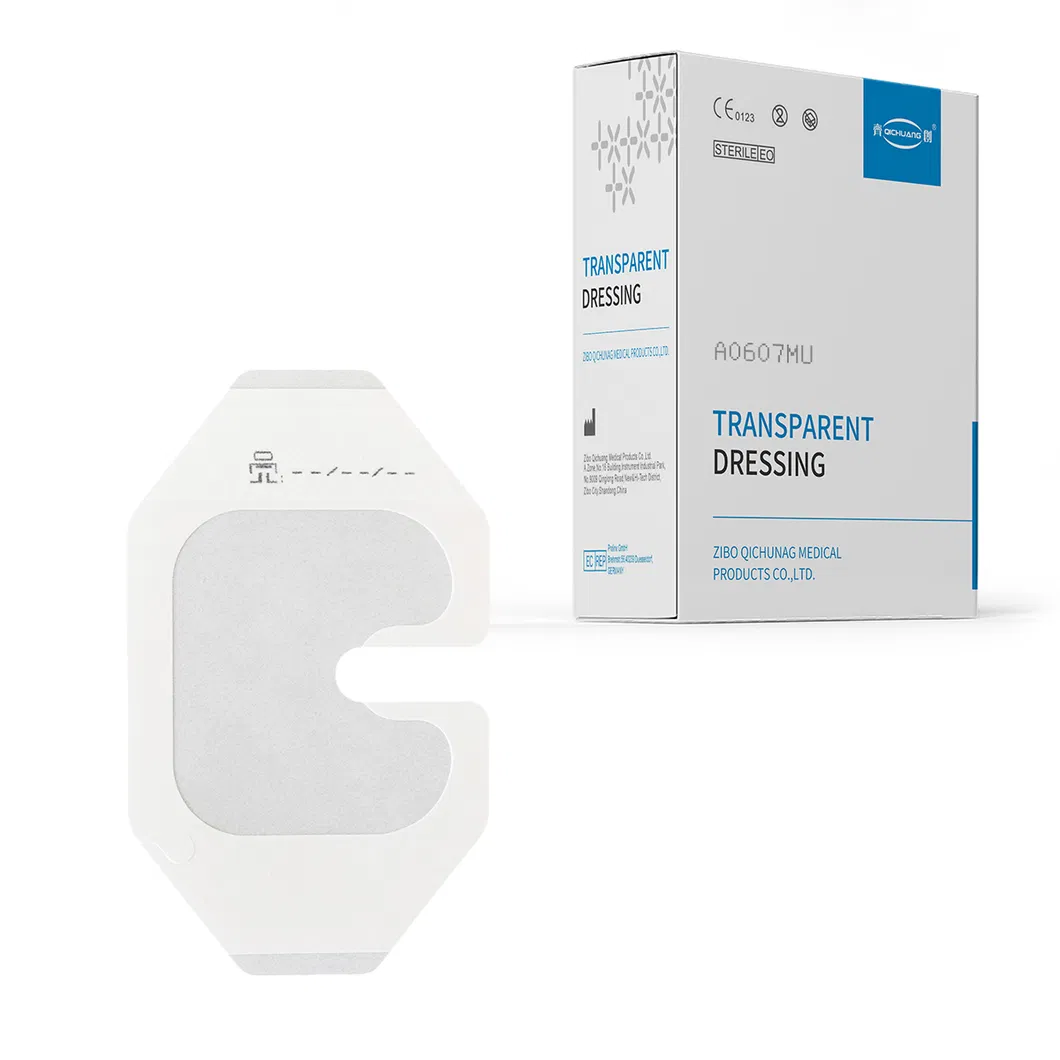
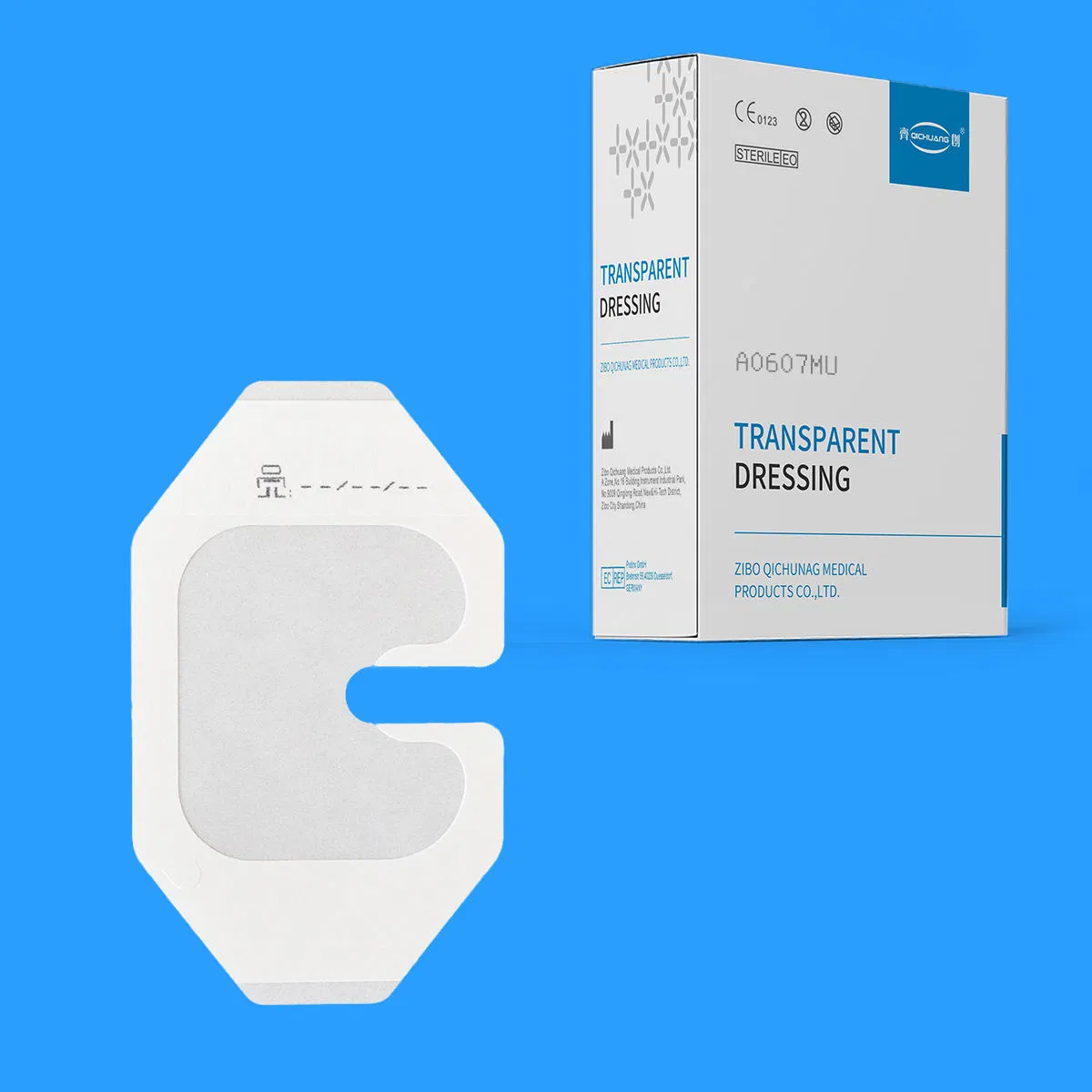
Individual Pack: Each piece is individually wrapped to ensure absolute sterility (Size: 90×135mm).
Bulk Supply: 100 pieces per box; 40 boxes per carton for convenient clinical storage.