Basic Information
Feature
Ventilation, Waterproof, Eco-Friendly
Function
Antibacterial, Bacteriostatic
Properties
Medical Adhesive & Suture Material
Material
PU Film, Non Woven, Adhesive
Application
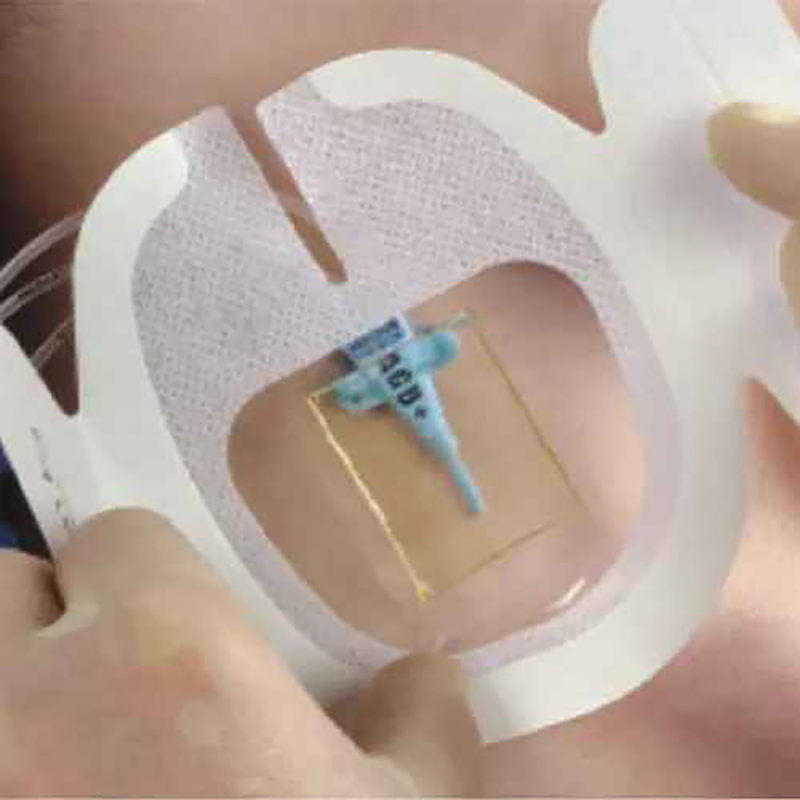
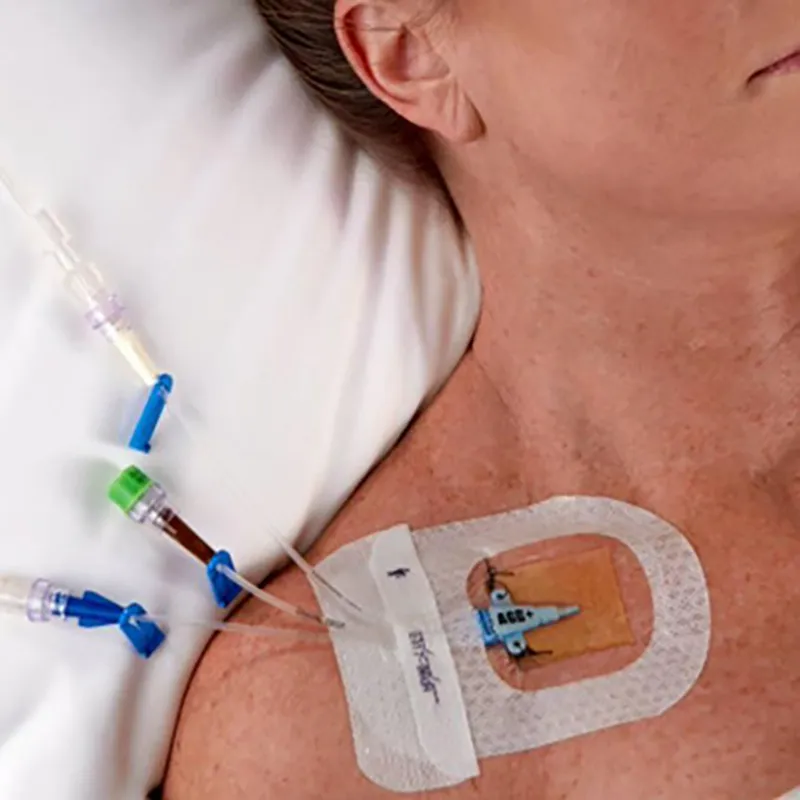
Fix CVC/Picc Central Venous Catheter
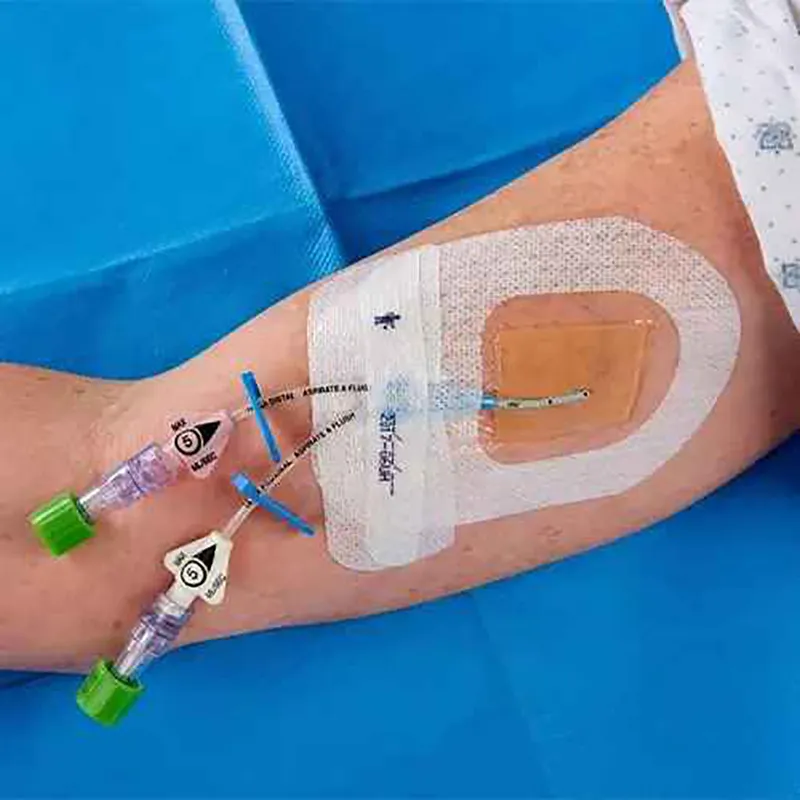
Product Overview
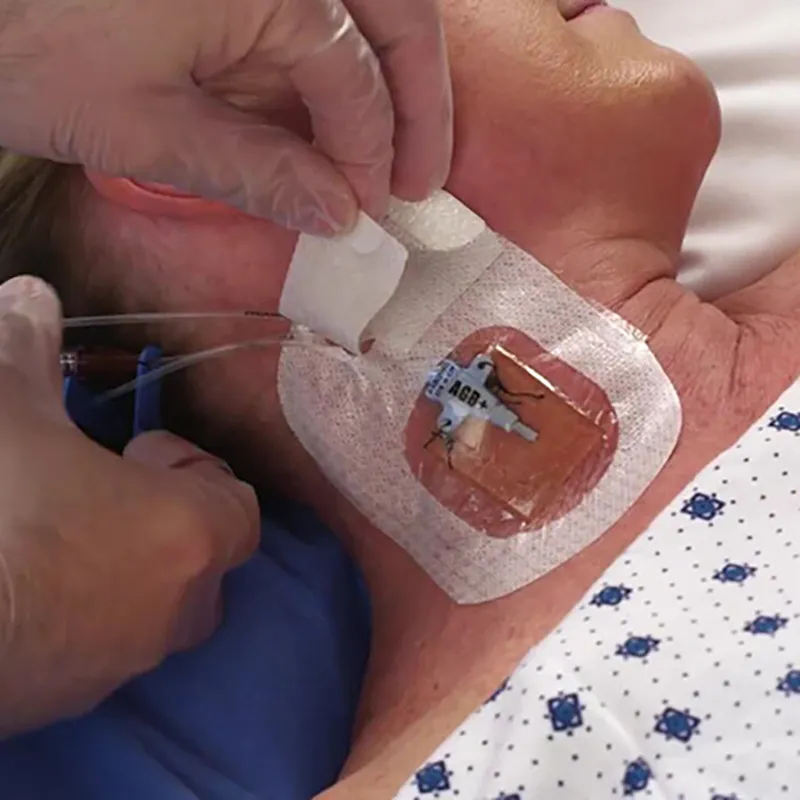
CHG I.V. Dressing is composed of a transparent dressing and a gel pad. The gel pad contains 2% chlorhexidine gluconate (CHG), which is a powerful antibacterial agent used to reduce skin colonies and inhibit microorganism regrowth.
Key Features
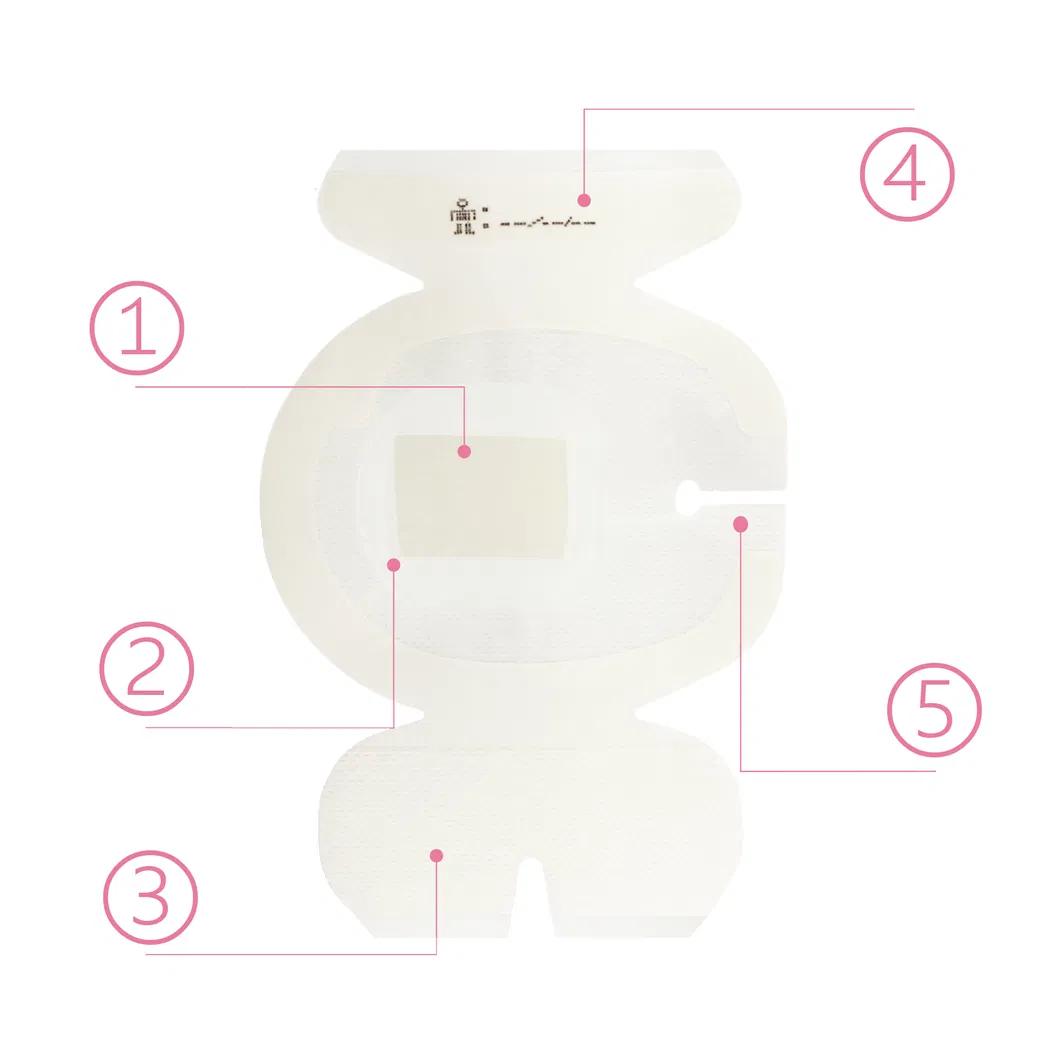
🛡️
CHG Gel Pad: Provides immediate and continuous antimicrobial protection.
💧
Breathable PU Film: Transparent and waterproof barrier against microbes and contaminants.
📍
Securement Notch: Specifically designed to keep catheters firmly in place.
📝
Documentation Strip: Includes a reinforcing tape strip for recording clinical data.
☁️
Non-Woven Reinforcement: Dot-coated border improves breathability and conforms to body contours.
Intended Usage
- Short peripheral I.V. and midline venous catheters
- Central venous catheters (Subclavian, Jugular, Femoral, PICCs)
- Dialysis and Arterial catheters
- Epidural catheters and other percutaneous devices
Primary Advantages
- Sterile barrier against liquids, bacteria, and viruses
- Continuous visibility of the I.V. insertion site
- Skin-friendly adhesive for patient comfort
- High breathability allowing skin to breathe freely
Specifications & REF Table
| REF Number |
Available Size |
| HT300-001 | 5x5.7cm |
| HT300-002 | 7x9cm |
| HT300-003 | 6x7cm |
| HT300-004 | 15x20cm |
| HT300-005 | 10x10cm |
| HT300-006 | 10x14cm |
Safety Warnings
1. Do not use on ulcerated wounds or unhealthy skin.
2. For external and disposable use only; avoid contact with mucous membranes.
3. Contraindicated for patients with known allergies to chlorhexidine gluconate (CHG).
4. If irritation or allergic reaction occurs, discontinue use immediately and consult a physician.

Frequently Asked Questions (FAQ)
Is the CHG IV dressing suitable for all skin types?
The dressing is hypoallergenic and skin-friendly; however, it should not be used by individuals with a known allergy to chlorhexidine gluconate.
Does this dressing provide protection against viruses?
Yes, the PU film creates a sterile, waterproof barrier that protects the insertion site from external contaminants, including liquids, bacteria, and viruses.
Can I see the insertion site while the dressing is applied?
Yes, the dressing is designed with a transparent film to provide continuous visibility for monitoring the IV site without removing the dressing.
What is the shelf life of the product?
The CHG IV dressing has a shelf life of 3 years from the date of manufacture when stored properly.
Are different sizes available for various catheter types?
Yes, we offer multiple sizes ranging from 5x5.7cm up to 15x20cm to accommodate short peripheral IVs, CVCs, and other percutaneous devices.
Is the product sterilized?
Yes, all dressings are individually packed and sterilized using EO (Ethylene Oxide) to ensure medical-grade safety.